Backlog and efficiency
October 9, 2015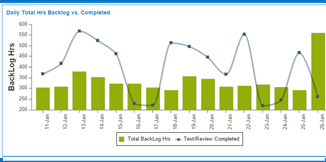
How to calculate backlog
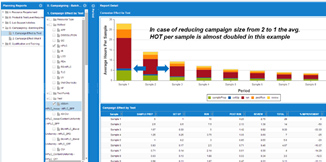
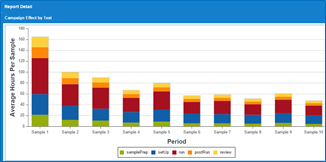
December 9, 2015We are often asked: if moving stability offshore makes sense and what is the expected savings? Furthermore, how do we calculate savings and what is the impact on the lab in the US vs. offshore? The simple answer is if the main target is reducing cost, then moving the stability samples offshore makes sense financially. Now let’s look at some of the aspects of moving these samples out of the commercial manufacturing site. Let’s assume the commercial lab is in US and the offshore site is set in India. For the US lab, moving the stability samples out, will reduce the opportunity for campaigning and for some low volume it may be a significant loss in terms of percentage, yet it is small volume. For a large volume product family, the impact is relatively minor as we usually run at a reasonable campaigning level. So here is the interesting scenario, although it does not affect the big picture: If there is a small volume of let’s say 2 samples – ONE stability and One Finished Product, the total testing is 15 hrs for the first sample, and 5 hrs for an additional sample. If the site remains in the US, then the expected Hands On Time is 20 hrs while if moved offshore, the Hands On Time becomes 15+15 = 30 hrs (total analyst and review hours). So in theory it makes no sense to move this product as we are losing money and this is true for most of the low volume products vs. high/medium products. The benefit of moving these products despite the inefficiency is to give the commercial lab site a focus on business and eliminate distractions. Obviously when considering managerial control and compliance, it may be desirable to keep the lab next to the SME / technical services team for support with any issues. Furthermore, using a good global LIMS and Scheduling system can provide the needed span of control. In conclusion, the cost aspect is almost always clear when comparing US analyst cost vs. India as example. However, the efficiency loss/gain is not as significant compared with the cost. On a final note, creating an offshore lab along with training, LIMS / Scheduling system implementation and compliance can be beneficial if carefully executed; however, it may cause a major headache and some writing by QA/QC.